Temple Utilizing Innovative Technology to Transport “Breathing Lungs” for Transplant from Donor to Recipient
Temple Recently Performed Pennsylvania’s First Post-FDA Approval Lung Transplant Using TransMedics Organ Care System for Lung
News October 21, 2020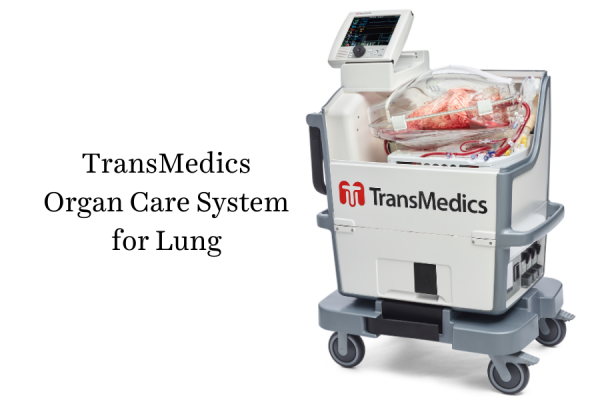 Temple University Hospital (TUH) recently became the first hospital in Pennsylvania to perform a breathing lung transplant using the TransMedics Organ Care System for Lung, or OCS™ Lung, following U.S. Food and Drug Administration (FDA) approval of the device for both standard and expanded criteria donor lungs. The OCS Lung is a portable technology designed to keep organs functioning and “breathing” in an environment that mimics the human body from the time they are removed from the donor until they are implanted in the recipient. The newest FDA approval expands the use of this technology to donor lungs initially deemed unacceptable for procurement and transplantation based on limitations of cold static preservation.
Temple University Hospital (TUH) recently became the first hospital in Pennsylvania to perform a breathing lung transplant using the TransMedics Organ Care System for Lung, or OCS™ Lung, following U.S. Food and Drug Administration (FDA) approval of the device for both standard and expanded criteria donor lungs. The OCS Lung is a portable technology designed to keep organs functioning and “breathing” in an environment that mimics the human body from the time they are removed from the donor until they are implanted in the recipient. The newest FDA approval expands the use of this technology to donor lungs initially deemed unacceptable for procurement and transplantation based on limitations of cold static preservation.
“Temple is able to serve a high volume of lung transplant patients, but tens of thousands of people in the U.S. are currently in need of lifesaving organs,” said Yoshiya Toyoda, MD, PhD, William Maul Measey Chair of Surgery at the Lewis Katz School of Medicine at Temple University (LKSOM) and Chief of Cardiovascular Surgery at TUH. “The hope is that advances in technology like the OCS Lung System will eventually afford more patients the opportunity to receive an organ transplant.”
The OCS Lung System allows medical teams to place donor lungs in a warm, sterile chamber that provides nutrient-rich and oxygenated blood to perfuse donor lungs while still at the donor hospital. Through a built-in ventilation circuit in the OCS Lung System, donor lungs will continue to breathe, allowing the medical team to recruit and optimize ventilation throughout the retrieval with no delayed or added time to the retrieval process. The current standard of care for preserving lungs prior to transplant is cold storage. However, the OCS Lung System reduces the time during which the donor lungs are cut off from blood and oxygen support outside of the body.
The donor lungs remain in the OCS Lung System perfused and ventilated during transport and until they are ready to be implanted in the organ recipient. In addition to preserving the lungs, the device measures and displays real-time data regarding the health of the organs.
Temple University Hospital performed more lung transplants than any other hospital in the nation in 2017, 2018 and 2019. Temple’s Lung Transplantation Program and the nationally recognized Temple Lung Center are comprised of physicians, nurses, therapists, pharmacists, social workers, laboratory technicians and administrative and support personnel who work around the clock to provide cutting edge medical and surgical care to patients with complex and advanced lung disorders.
“The Temple Lung Center is able to offer its patients access to some of the most advanced treatments and clinical trials available in the areas of prevention, diagnosis and treatment of complex lung disease,” said Francis Cordova, MD, Professor of Thoracic Medicine and Surgery at LKSOM and Medical Director of the Lung Transplantation Program at TUH.
“Temple’s Lung Transplantation Program is one of the nation’s most active, experienced and innovative, regularly providing lifesaving transplants in some of the most challenging cases,” said Norihisa Shigemura, MD, PhD, Professor of Surgery at LKSOM and Surgical Director of the Lung Transplantation Program at TUH.
“This innovative technology is state-of-the-art in its ability to preserve the health of the organ in such a small chamber and to ventilate in real-time before the organ is implanted in patients, says M. Abul Kashem, MD, PhD, a Research Faculty and Assistant Professor of Surgery at LKSOM who oversees Temple’s use of the OCS Lung. “Our team is looking forward to seeing the future results of such device-based lung transplant technology that benefits waiting list patients.”
Dr. Kashem, along with the surgical and medical team, will analyze survival outcomes for cases in which the OCS Lung technology is utilized, and will also assess long-term benefits for patients.
Editor’s Note: Neither Dr. Toyoda, Dr. Cordova, Dr. Shigemura, Dr. Kashem nor any member of their immediate families has financial interest in TransMedics.
